|
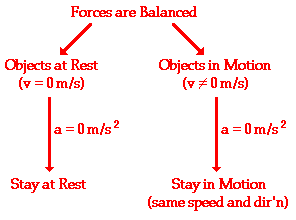
Newton's first law of motion is often stated as: An object at rest stays at rest and an object in motion stays in motion with the same speed and in the same direction unless acted upon by an unbalanced force. Two Clauses and a Condition There are two clauses or parts to this statement - one that predicts the behavior of stationary objects and the other that predicts the behavior of moving objects. The two parts are summarized in the following diagram. The behavior of all objects can be described by saying that objects tend to "keep on doing what they're doing" (unless acted upon by an unbalanced force). If at rest, they will continue in this same state of rest. If in motion with an eastward velocity of 5 m/s, they will continue in this same state of motion (5 m/s, East). If in motion with a leftward velocity of 2 m/s, they will continue in this same state of motion (2 m/s, left). The state of motion of an object is maintained as long as the object is not acted upon by an unbalanced force. All objects resist changes in their state of motion - they tend to "keep on doing what they're doing."
There is an important condition that must be met in order for the first law to be applicable to any given motion. The condition is described by the phrase "... unless acted upon by an unbalanced force." As the long as the forces are not unbalanced - that is, as long as the forces are balanced - the first law of motion applies.
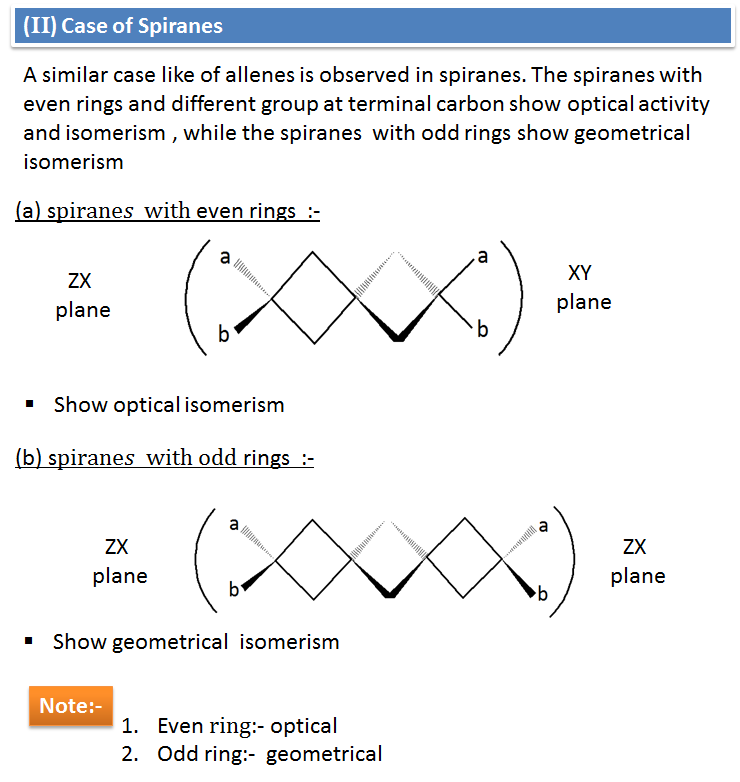
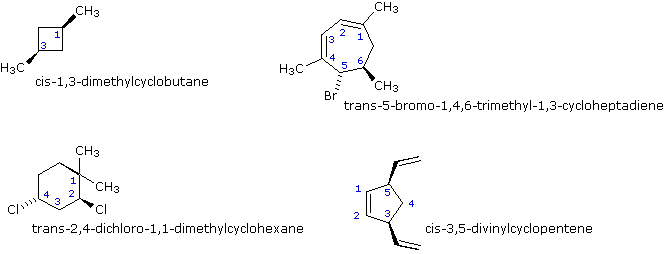
Activation energy for the rotation about carbon–carbon double bond is very high (250KJ mol–1). This is the contribution of bond to the total C = C bond energy of 605 kJ mol–1. Because of high activation energy, rotation about C = C is restricted. As a result, two spatial arrangements of groups is possible at C = C. They are named as cis and trans isomers or geometrical isomers.
i) Cis-isomer is more polar than the trans-isomer because in it, the individual dipoles do not cancel each other. ii) Cis-isomer has greater boiling point than the trans isomer due to greater polarities and consequent stronger attractive forces. iii) Trans-isomer has higher melting point than the cis-isomer because its molecules are more closely packed in solid state due to symmetry. iv) Trans-isomer is more stable than the cis-isomer due to less repulsive forces. v) Cis-isomer has higher solubility in polar solvents. vi) Cis-isomer has higher density, refractive index, dipole moment and heat of combustion than its trans isomer. |
AuthorSuresh Gdvm ArchivesCategories |
||||||
- About Us
- Biology
-
Chemistry
-
Class XI - Chemistry
>
-
Class XII - Chemistry
>
- Solid State
- Solutions
- Electrochemistry
- Chemical Kinetics
- Surface Chemistry
- Processes of Isolation of Elements
- The p-block Elements
- The d- and f- block Elements
- Coordination Compounds
- Haloalkanes and Haloarenes
- Alcohols, Phenols and Ethers
- Aldehydes, Ketones and Carboxylic Acids
- Organic compounds containing Nitrogen
- Biomolecules
- Polymers
- Chemistry in Everyday Life
-
Class XI - Chemistry
>
- Mathematics
- Physics
- Science 360 Blog
- Science Info
- Contact Us

 RSS Feed
RSS Feed

